
Figure 1: Researchers out of Harvard Medical School find that reprogramming OSK transcription factors interferes with age-related gene expression, ultimately posing as a potential method of counteracting age-related symptoms
Turning back the biological clock is a challenge that scientists around the world have chosen to undertake in the past several decades. Telomere shortening, molecular inflammation, and oxidative stress are just a few of the proposed contributors towards aging (Chung et al., 2008; Blasco, 2007; Liguori et al., 2018). In a recent study out of Harvard Medical School, epigenetics has now been proposed as another factor that interferes with age-related gene expression, leading to tissue decline and a decrease in regenerative characteristics (Lu et al., 2020).
DNA methylation patterns have been known to correlate to biological age, although its mechanisms remain unclear (Johnson et al., 2015). These Harvard researchers found that the OCT4, SOX2, KLF4 and MYC (OSKM) genes may reprogram somatic cells to become pluripotent stem cells in culture, resetting their methylation ages and thus potentially counteracting aging (Lu et al., 2020). They subsequently investigated whether mammalian cells could retain a youthful epigenetic profile and exhibit increased longevity.
To reset the epigenome without altering cellular identity, Lu et al. (2020) removed MYC, an oncogene, and studied the effects of OCT4, SOX2, and KLF4 (OSK) transcription factors in mice and how they influenced known age-related genes such as Lmnb1, Chaf1b, and the H2a and H2b families (Yang et al., 2019). They found that these transcription factors stimulated a youthful mRNA profile with no loss of cellular function, as well as no increase in tumor incidence or wellbeing.
This discovery led to further work in one of the first systems to lose its regenerative capacity, the central nervous system (CNS) (Silver, Schwab, & Popovich, 2014). After supplementing retinal ganglion cells (RGCs) of the CNS with OSK transcription factors, it was found that OSK treatment induced axonal regeneration, with aging playing a limited diminishing role (Lu et al., 2020). Furthermore, Lu et al. (2020) discovered that injury-induced global DNA methylation alterations were reversed by OSK transcription factors, with an increased localized expression in light-detection and synaptic-transmission genes.
This begged further inquiry into the supplementary effects of OSK on vision, namely for patients with glaucoma, a leading cause of blindness, and age-related vision deterioration (Choquet et al., 2020). In OSK-supplemented, glaucoma-induced mice, researchers found a significant increase in visual acuity in comparison to the pre-treatment function baseline, indicative of a positive effect of OSK gene expression on vision-loss reversal (Lu et al., 2020). OSK induction also reversed the DNA methylation aging signature, indirectly recruiting TET enzymes. These enzymes, namely Tet1 and Tet2, assist in the improvement of RGC function and are therefore a key player in vision restoration for old mice (Lu et al., 2020).
Throughout the study, OSK transcription factors were demonstrated to play a counteracting role in aging, and have potential to reverse some of the currently incurable symptoms associated with longevity, such as neurocognitive decline, frailty, and inflammation (Park & Reuter-Lorenz, 2009; Fulop et al., 2010). Epigenetic reprogramming may be a means of reversing age-related decline in humans, and should be carefully investigated to yield a better understanding of the biological process that aging entails. Eventually, these findings may lead to the creation of therapeutics aimed at age-related diseases.
References
Blasco, M. A. (2007). Telomere length, stem cells and aging. Nature Chemical Biology, 3(10), 640–649. https://doi.org/10.1038/nchembio.2007.38
Choquet, H., Wiggs, J. L., & Khawaja, A. P. (2019). Clinical implications of recent advances in primary open-angle glaucoma genetics. Eye, 34(1), 29–39. https://doi.org/10.1038/s41433-019-0632-7
Chung, H. Y., Cesari, M., Anton, S., Marzetti, E., Giovannini, S., Seo, A. Y., … Leeuwenburgh, C. (2008). Molecular inflammation: Underpinnings of aging and age-related diseases. Ageing Research Reviews, 8(1), 18–30. https://doi.org/10.1016/j.arr.2008.07.002
Fulop, T., Larbi, A., Witkowski, J. M., Mcelhaney, J., Loeb, M., Mitnitski, A., & Pawelec, G. (2010). Aging, frailty and age-related diseases. Biogerontology, 11(5), 547–563. https://doi.org/10.1007/s10522-010-9287-2
Johnson, A. A., Akman, K., Calimport, S. R., Wuttke, D., Stolzing, A., & Magalhães, J. P. D. (2012). The Role of DNA Methylation in Aging, Rejuvenation, and Age-Related Disease. Rejuvenation Research, 15(5), 483–494. https://doi.org/10.1089/rej.2012.1324
Liguori, I., Russo, G., Curcio, F., Bulli, G., Aran, L., Della-Morte, D., … Abete, P. (2018). Oxidative stress, aging, and diseases. Clinical Interventions in Aging, Volume 13, 757–772. https://doi.org/10.2147/cia.s158513
Lu, Y., Brommer, B., Tian, X., Krishnan, A., Meer, M., Wang, C., … Sinclair, D. A. (2020). Reprogramming to recover youthful epigenetic information and restore vision. Nature, 588(7836), 124–129. https://doi.org/10.1038/s41586-020-2975-4
Park, D. C., & Reuter-Lorenz, P. (2009). The Adaptive Brain: Aging and Neurocognitive Scaffolding. Annual Review of Psychology, 60(1), 173–196. https://doi.org/10.1146/annurev.psych.59.103006.093656
Silver, J., Schwab, M. E., & Popovich, P. G. (2014). Central Nervous System Regenerative Failure: Role of Oligodendrocytes, Astrocytes, and Microglia. Cold Spring Harbor Perspectives in Biology, 7(3). https://doi.org/10.1101/cshperspect.a020602
Yang, J.-H., Griffin, P. T., Vera, D. L., Apostolides, J. K., Hayano, M., Meer, M. V., … Sinclair, D. A. (2019). Erosion of the Epigenetic Landscape and Loss of Cellular Identity as a Cause of Aging in Mammals. Preprint (Courtesy of BioRxiv). https://doi.org/10.1101/808642
Related Posts
Universal Donors: A New Enzymatic Approach to Blood and Organ Matching
Figure: In a 2019 study, researchers used a microbial sample...
Read MoreYour Personality Might Say Less About Your Brain, And More About Your Microbiome
Figure 1: A scanning electron micrograph of Enterococcus faecalis, a bacterium...
Read MoreDirecting Neural Stem Cells to Repair Damage
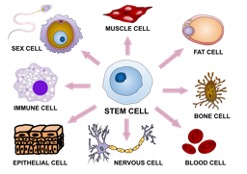
Figure 1: This depicts the ‘undifferentiated’ nature of pluripotent stem...
Read MoreBrandon Yu

Comments are closed.